Havana, Jun 14 (Prensa Latina)
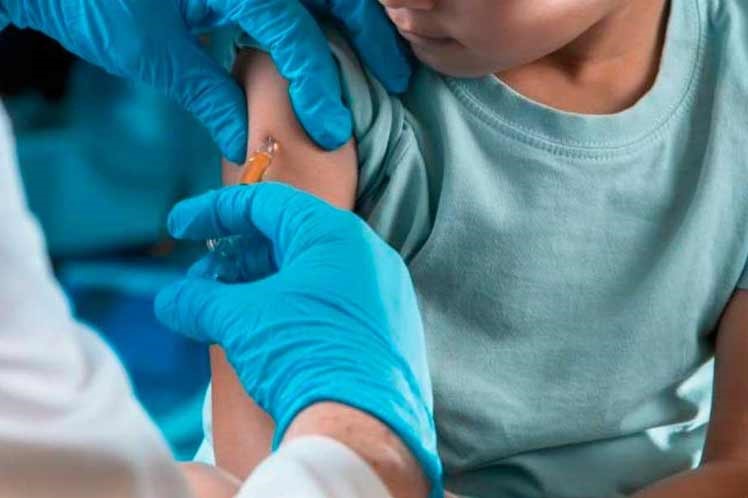
Cuba has begun on Monday phases I/II of the clinical trials of its Soberana 02 and Soberana Plus Covid-19 vaccines on Children and teenagers, with two doses of the former and one of the later.
Approved on June 10 by the Center for State Control of Medicines, Equipment and Medical Devices (CECMED), these trials that will take place at the Juan Manuel Marquez Pediatric Hospital in Havana will be sequential, open and adaptive for three to 18 years of age.
To carry this out, 350 volunteers of pediatric ages will be divided into two age groups: from 12 to 18 and from three to 11.
This stage aims to assess the safety, reactogenicity, and immunogenicity of the prophylactic vaccine candidates designed by the Finlay Institute of Vaccines (IFV) in Havana, at 28-day periods, a scheme already applied to the adult population.
Dr. Meiby de la Caridad Rodriguez, director of Clinical Research at IFV, explained that after every dose, children will remain under surveillance for an hour at the vaccination center, then they will have to go again every 24, 48 and 72 hours so that the specialists verify if they had adverse effects related to the vaccine.
mh/iff/jf/cdg

Comments are closed.